Lifyorli (relacorilant, Corcept Therapeutics) received its first approval on March 25th based on the results from the phase 3 ROSELLA trial, which looked at the Corcept agent as an add-on to nab-paclitaxel in platinum-resistant ovarian cancer (PROC) patients who have received prior bevacizumab. [1] These patients have limited treatment options and a mortality rate in the first year of platinum-resistant disease that is 16 times higher than that of their age-matched peers. Recent FDA approvals have emerged in subsets of PROC patients with actionable biomarkers, but little options remain for patients without them. As a result, these patients will typically receive non-platinum chemotherapy, such as paclitaxel, pegylated liposomal doxorubicin, or topotecan. [1]
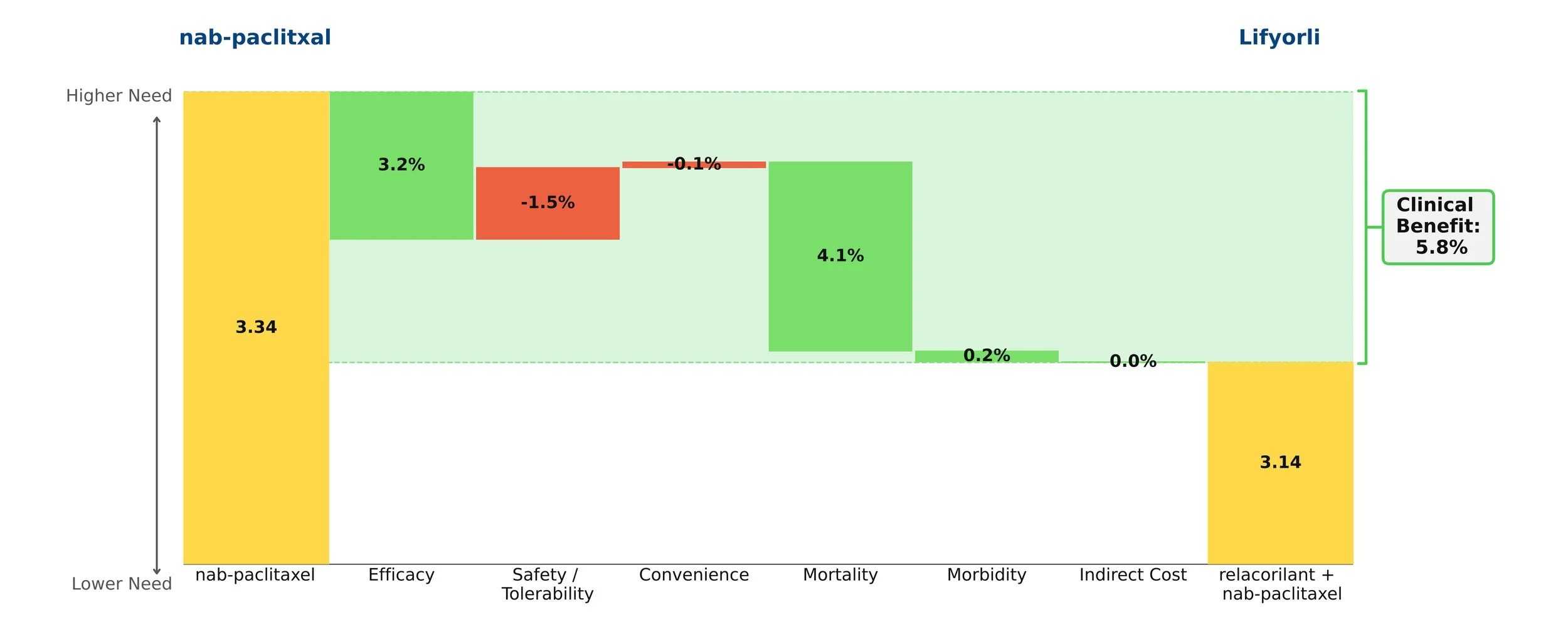
When added onto nab-paclitaxel, Lifyorli offered over 4 additional months of survival compared with nab-paclitaxel alone. (16.0 vs 11.9, HR = 0.65) [1] This substantial benefit helps to meaningfully address the most substantial unmet need of this disease. However, the regimen did not offer the same magnitude of improvement in progression-free survival and overall response, only scoring one additional month of PFS (6.5 vs 5.5) and not achieving a statistically significant difference in the secondary endpoint of overall response rate (36.9% ORR vs 30.1%, p=0.17). [2] Together, these attributes make up an interesting efficacy profile, as an OS improvement more than 4x the PFS improvement of a given regimen is highly uncommon.
When taking into account the modest drawback of increased side effects, Lifyorli achieves a solid 5.8% clinical innovation when compared to nab-paclitaxel. While this is not a blockbuster score, it is no doubt a meaningful efficacy improvement that will make this drug competitive.
Figure 1: Drivers of Clinical Benefit