Narcolepsy is a rare neurological disorder that affects an estimated 200,000 Americans [1], and about half of those people have narcolepsy type 1 (NT1) [2]. While both NT1 and NT2 cause sleep attacks and excessive daytime sleepiness, NT1 also causes cataplexy: episodes of sudden muscle weakness often triggered by intense emotions. Not only does NT1 severely impact quality of life, but it can also be dangerous when cataplexy episodes or sleep attacks occur during activities such as driving or operating machinery [3].
In February 2026, the FDA accepted the NDA for oveporexton, Takeda’s investigational agent, and granted it Priority Review. Instead of simply managing symptoms, oveporexton is a potentially first-in-class OX2R-selective agonist that targets the orexin deficiency that causes NT1 [4].
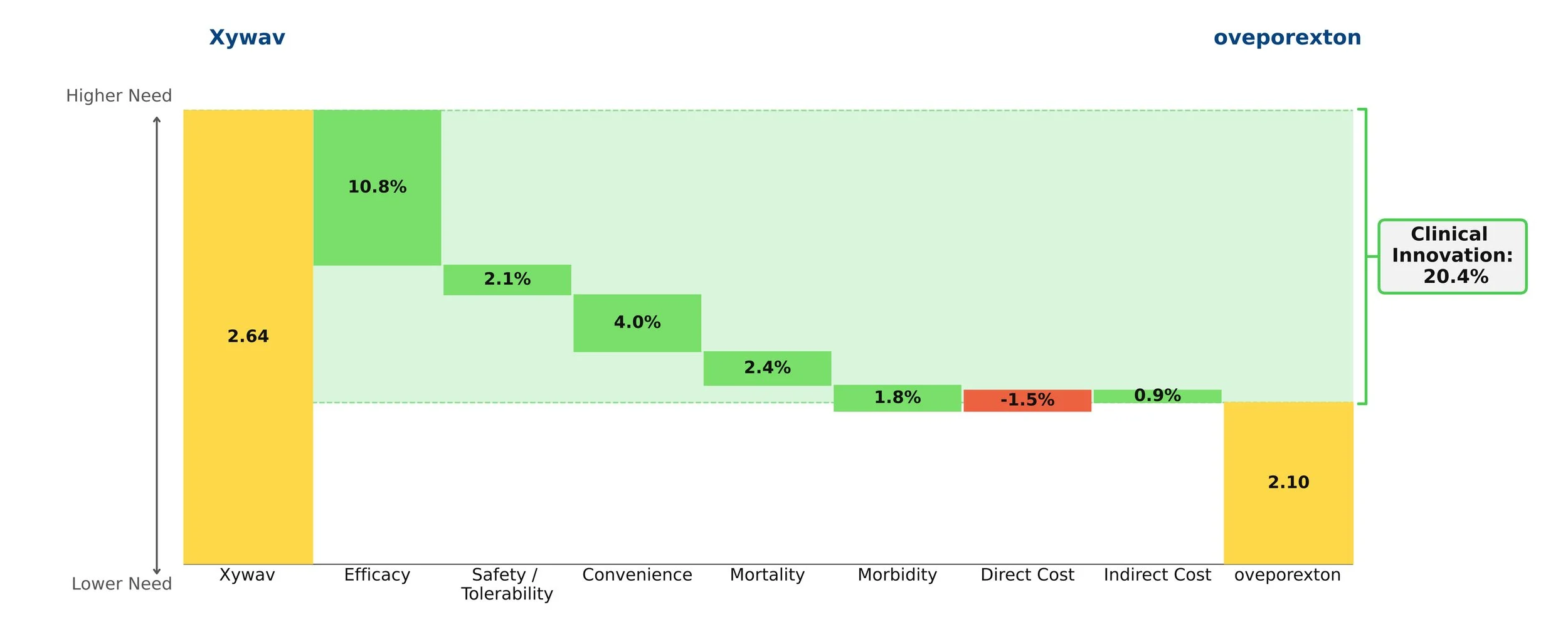
Oveporexton yields an exceptional clinical innovation score of 20.4% over standard-of-care Xywav when it is priced at a modest premium. Based on currently available data, we view oveporexton as a more efficacious and convenient option, and, if launched at this price point, expect it will take a commanding lead of the NT1 market.